The salt spray test in accordance with DIN EN ISO 9227 (often referred to simply as the salt spray test) is used to evaluate the quality of surface pretreatment and coatings on metals. During testing, the specimen parts are exposed to precisely defined, extreme conditions. This makes it possible to draw indirect conclusions about the effectiveness of corrosion protection. Read on to learn when a salt spray test is required and how it is carried out.
Application of DIN EN ISO 9227 (Corrosion tests in artificial atmospheres – Salt spray tests)
Corrosion preventatives enable the use of base metals in a wide variety of components. Its effectiveness determines the service life of machines and systems, vehicles, and structures. Manufacturers of such products expect their suppliers to deliver parts that meet defined quality standards. To verify this quality, clear specifications and criteria are required. These are often summarized in the internal standards of major manufacturers. Within this framework, a salt spray test in accordance with DIN EN ISO 9227 may be required. Alternatively, this requirement may be agreed upon contractually. The standard specifies how the test apparatus must be designed, which substances are used, and under what conditions the test is completed . All other specifications are defined by the company requesting the test from its suppliers. This includes the nature and size of the test specimens, specimen preparation, test duration, and the required results.
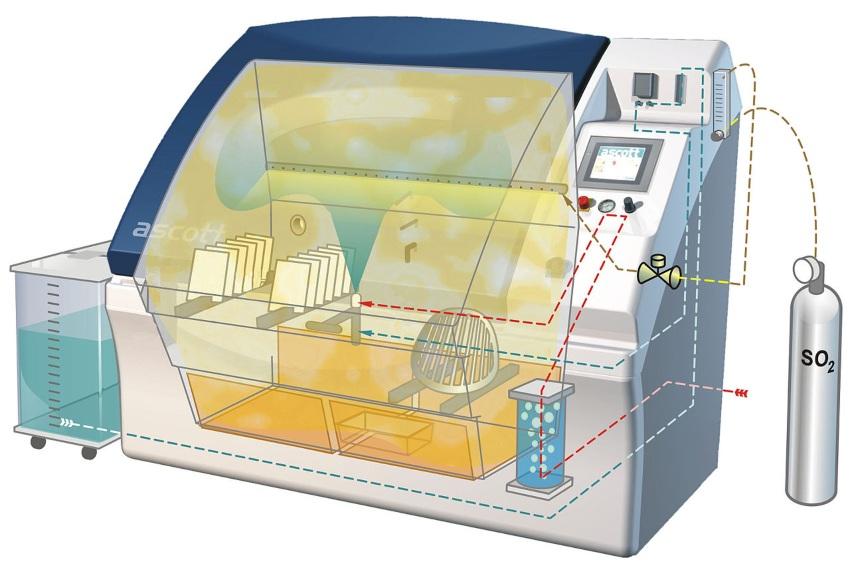
Artificial, corrosion-promoting atmosphere
The salt spray test is conducted in a chamber that is continuously supplied with a finely atomized salt solution via nozzles. The standard specifies three variants of the salt solution, each producing a different level of corrosive attack. The neutral salt spray test (NSS test) uses a 5% aqueous sodium chloride solution. A slightly more aggressive method is the acetic acid salt spray test (AASS test), in which acetic acid is added to the sodium chloride solution. The third variant is the copper-accelerated acetic acid salt spray test (CASS test). In this case, the solution contains sodium chloride, acetic acid, and copper(II) chloride dihydrate. DIN EN ISO 9227 specifies in detail how these solutions should be prepared. It also defines required spray volumes, pH values, and temperatures to be used during testing. These specifications, in turn, dictate chamber equipment requirements for heating, dosing, measurement, and control.

Arrangement of test specimens in the chamber
To obtain reproducible results, the test specimens in a salt spray test must be exposed solely to the corrosive action of the artificial atmosphere. Other influences, such as contact with other metals or standing moisture on the surfaces, must be excluded. For this reason, there are precise requirements regarding the materials used for the chamber and the racks holding the test specimens. The positioning of the specimens can also influence the results of the salt spray test. The following rules apply:
- as few contact points as possible between the test specimen and the rack or suspension
- no contact between different test specimens
- arrangement of the test specimens at an angle of 20° from the vertical so that condensate separating from the mist can drain freely
- condensate must not drip onto other test specimens
Evaluation and documentation of salt spray test results
The customer specifies which variant of the salt solution is to be used, how long the coating or surface pretreatment of the workpieces must withstand the defined conditions, and which surface changes are permissible. In addition, the customer determines the required characteristics of the test specimens.
After completion of the test period, the specimens tested in the salt spray test are rinsed with distilled water and dried. This is followed by a visual inspection for surface changes. If the salt solution penetrates the conversion coating formed during surface pretreatment, corrosion products form on the base material. This occurs at points where the layer is damaged or contains contaminants. This is evident from blistering under paint and coating layers or from the color and extent of corrosion products. Rust on iron is typically reddish-brown, while zinc forms white, spot-like corrosion products.

It is often of interest to determine how far paints and coatings used for corrosion protection can be undermined by aggressive media in damaged areas. In such cases, the coating is simply scratched, either randomly or according to a defined pattern, before the salt spray test. After the test, it is determined whether and to what extent the coating has detached at the scratched areas.
All observations are carefully documented together with the test duration, the measurement data from the salt spray test, and a description of the test setup, are recorded in a test report. The observations are then compared with the customer’s requirements. If the changes fall within the specified tolerance range, the test is considered passed.

Informative value of salt spray test results
The salt spray test provides results that can only be used to assess the quality of a surface coating. It allows comparable coating systems on identical base materials to be evaluated. The conditions of salt spray testing differ significantly from the real environmental conditions that corrosion protection must withstand in actual use. For this reason, the test can only provide indirect indications of the corrosion behavior of the tested components. In simplified terms: the coating is acceptable, has proven itself in practice, and therefore also protects against corrosion. This applies primarily to organic coatings such as paints and lacquers, as well as to anodically produced oxide layers (e.g., anodized aluminum). By contrast, the informative value is limited for metallic coatings applied by electroplating or hot-dip galvanizing.
 Kluthe Magazine
Kluthe Magazine
