Most metals used to manufacture a wide variety of products occur in nature only in the form of chemical compounds known as ores. Only after they are released from these compounds are they available in metallic form. Left unprotected, these metals will corrode. The materials react with substances from their environment as they attempt to return to their original state. Read on to learn which types of corrosion occur and how the resistance of metallic materials can be increased through surface treatment processes.
Criteria for distinguishing types of metal corrosion
Corrosion damages the material. Without countermeasures, many metal products would have a very short service life. Suitable solutions for corrosion protection can only be identified if the causes and mechanisms of corrosion processes are considered. Individual metals and alloys react differently to corrosive attack by environmental media. Both the chemical elements involved, and the material structure formed during solidification of the melt play a role in the type of corrosion. In addition, prior metal processing, and the design of the component influence corrosion behavior. Finally, differences arise in the way material degradation spreads, whether limited to the metal surface or extending into the depth of the material. Based on these criteria, distinct types of metal corrosion are classified to facilitate targeted prevention.
Types of corrosion based on reaction mechanisms
Formation of local galvanic cells
Understanding corrosion starts with the atomic structure of metals. Atoms are arranged in regular lattices. The outer electrons of the atomic shells are freely movable within these lattices. Their absence from individual atomic shells allows the positive charge of the atomic nuclei to dominate. Strictly speaking, these lattices consist of ions that respond to electric fields. When moisture is present, galvanic cells form. Such corrosion processes correspond in their processes to galvanic coating or the electrolysis of water. A small electric current causes metal ions to dissolve at locations where there is an electron deficiency (anode) and combine with oxygen from moisture. At locations with an excess of electrons (cathode), gaseous hydrogen is produced, which immediately reacts with oxygen in the air to form water. The material areas in which these processes take place usually have only a very small spatial extent. This results in the term local galvanic cells.
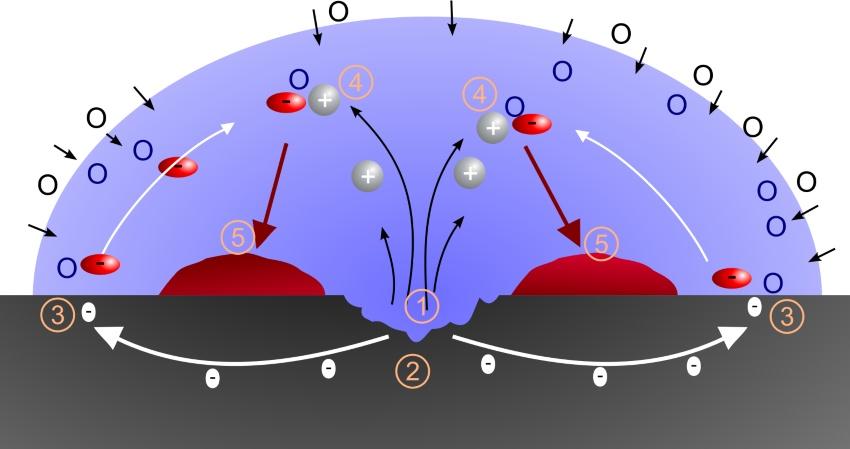
Contact corrosion
When different metals come into contact with each other, an electrical potential difference can be observed between them. An electric field forms. When metals are arranged according to the magnitude of these potential differences, the electrochemical series results. Hydrogen is arbitrarily placed in the middle of this series. Metals that are more positive in the electric field are considered noble, while those that are more negative are considered base. The greater the distance between metals in the series, the greater the potential difference and thus the electric current that flows when a closed circuit is formed. This current consists of ions from the less noble metal, which detach from their lattice, migrate toward the more noble metal, and deposit there. These types of corrosion can be suppressed by using identical materials within a system or by careful electrical isolation.

Chemical oxidation
In addition to electrochemical processes, some types of corrosion are caused by direct chemical reactions. When certain dry gases, salts, bases, or acids make contact with metal surfaces, they form molecules whose stability is based on shared electrons. These corrosion processes are known as redox reactions. The metal receives electrons for shared use and is oxidized. The reaction partner provides these electrons and is reduced. These processes occur more intensely at high temperatures. Substances that can trigger such behavior include oxygen, hydrogen sulfide, ammonia, chlorine, and ammonium salts. These types of corrosion can be avoided through the use of resistant materials or suitable coatings.

Biocorrosion
Biocorrosion is another important type of metal corrosion. In this form of corrosion, the substances responsible for the corrosive attack originate from the metabolic processes of living organisms. Microorganisms, fungi, lichens, plants, and animals that colonize metal surfaces produce an almost inexhaustible range of organic substances that can form chemical compounds with metals. These include various organic acids, such as formic acid, acetic acid, uric acid, and citric acid. In addition, metabolic by-products such as hydrogen sulfide and nitrogen oxides can form, which under certain conditions react with water to form sulfuric or nitric acid.
Types of metal corrosion based on propagation mechanisms
Uniform corrosion
In this type of corrosion, the electrochemical conversion of the material occurs evenly across the metal surface. Depending on the material used, thin passive layers quickly form under the influence of atmospheric oxygen and moisture, protecting the underlying material from further degradation. This is the case, for example, with chromium, tin, and stainless steel. However, the oxide layers on many iron-based materials are loose and porous. Without corrosion protection, material degradation continues until the material is completely consumed.

Pitting corrosion
Pitting corrosion is one of the electrochemical types of corrosion. It occurs primarily when passive layers on metal surfaces contain defects. Material degradation is intensified by the influence of chloride or bromide ions. These ions occupy the defects and prevent oxygen from reaching the surface, which would otherwise repair the passive layer. As a result, localized damage initially forms and continues to penetrate deeper into the base metal.

Crevice corrosion
If a metal structure contains narrow crevices, for example at overlapping edges, attached components, or interrupted weld seams, the concentration of surrounding substances inside the crevice differs from that outside. This concentration difference creates a potential difference similar to that between different metals. As a result, electrochemical reactions occur within the crevice.
Additional types of metal corrosion
Depending on material composition, microstructure, mechanical stresses during metal processing or operation, and operating temperature, numerous additional types of corrosion are distinguished. Examples include:
- Intergranular corrosion
- Stress corrosion cracking
- Corrosion fatigue
- Undercutting corrosion
- High-temperature corrosion
- Knife-line corrosion
- Erosion corrosion
- Stray current corrosion
Damage caused by these types of corrosion is primarily prevented through careful material selection and corrosion-resistant design.

Corrosion protection through substances and surface engineering processes
Corrosion inhibitors for protection during processing and transport
Corrosion inhibitors protect metal materials from corrosive attack during processing and transport. These substances are added to process fluids such as metalworking fluids or cleaning agents, or are included in packaging. Corrosion inhibitors work through two main mechanisms. Some protect metal surfaces by covering vulnerable areas of the material. Others react with corrosive media and thereby neutralize their effect.
Surface treatment to protect metal products from corrosion
The most common types of metal corrosion can be suppressed through appropriate coatings. For this purpose, a wide range of processes are used in surface treatment and surface pretreatment.

Examples include:
- Formation of conversion coatings (conversion processes)
- Painting
- Powder coating
- Electroplating
- Galvanizing
These coatings prevent environmental media from reacting with the metal surfaces.
 Kluthe Magazine
Kluthe Magazine

